New possibilities, as well as unexpected and unwanted effects, arise during different phases of drugs development. Simulx allows to create a new project from scratch, which gives freedom in simulating new scenarios. Moreover, it creates easily a space for testing new hypothesis. This helps to explore the full potential of drugs, and better prepare for their possible failures.
To create a new project from scratch:
- Select “New project” after opening the Simulx.
- Browse, or write new, mlxtran model (see here for details about the model structure).
- Use Definition tab to specify simulation elements required by the model. Explore in the Exploration tab the effects of treatments and parameters. Build a simulation scenario in the Simulation tab and run the simulation.
Demo project: 1.overview/newProject_TMDDmodel
The following example shows how to build “from scratch” a simulation that aims at comparing different treatments among two populations: healthy volunteers and patients with rheumatoid arthritis. It uses a TMDD model for a ligand and receptor concentrations. In addition, the synthesis of the receptor depends on the population type – lower for healthy subjects. All simulation elements are user-defined. The basic assumption is that a treatment is effective if the free receptor stays below 10% of the baseline. The goal is to test if the dose levels effective on healthy volunteers allow rheumatoid arthritis patients to reach the efficiency target.
Model
When building a project from scratch, loading a model is mandatory and has to be done in the first place. It sets automatically the structure of a simulation based on the model type and its elements.
- Structural model is a TMDD model with the QE approximation, two compartments and oral administration.
- Statistical model defines an effect of the patient state (healthy or with RA) on the synthesis rate of the receptor. It also includes the power law relationship between the weight covariate and the volume of the central compartment.
Covariates and their transformations are described in the [COVARIATE] block, the statistical model of the individual parameters is in the [INDIVIDUAL] block, while the [LONGITUDINAL] block contains the structural model with output definitions.
Definition of the scenario elements:
To guide building a project from scratch, Simulx defines default simulation elements automatically using information from the loaded model. These elements can be removed or modified, as well as new elements can be created. In this example, the definition includes the following new elements used in the exploration and simulation.
- Pop.Param.: Values come from an external table with at least two mandatory rows. The first row contains names of all population parameters as used in the model, while the second has the parameter values.
- Indiv.Params.: Individual parameters appear in the exploration tab. In this example, they are equal to population parameters in order to explore the effect of parameters on the model predictions for a typical individual.
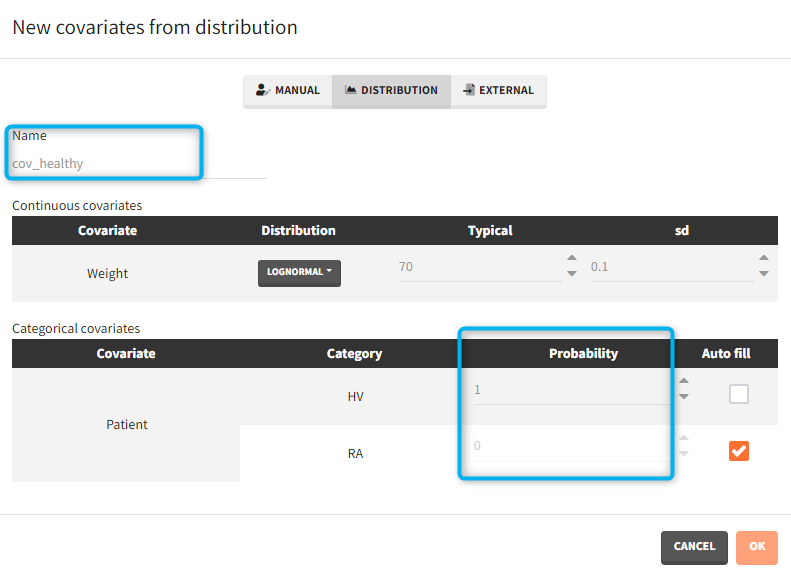
- Covariates: The aim of this simulation is to compare two populations (healthy and with rheumatoid arthritis). There are two covariate elements with different values of the “patient” covariate, but the same “weight” covariate.
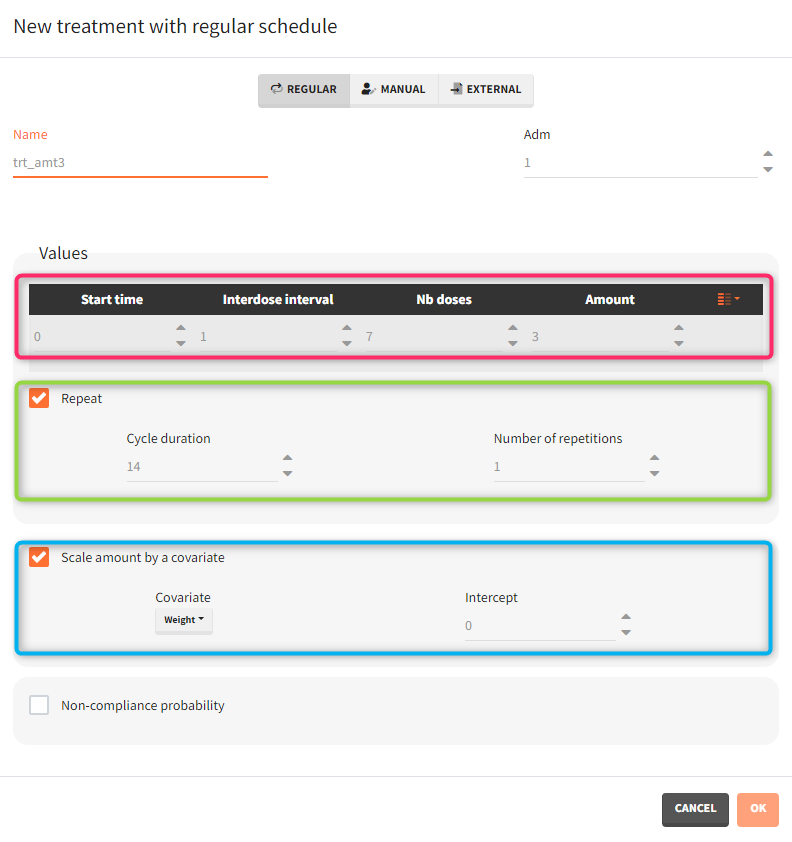
- Treatment has two cycles (green frame). In each cycle patients receive an oral dose scaled by their weight (blue frame) once per day for 7 days (red frame) followed by 7 days without a treatment. Three different dose levels: 3mg/kg, 6mg/kg and 12mg/kg require three separate treatment elements . An option “duplicate” allows to clone a treatment, and then change only the name and the amount. Note that the “View” option has the scaling formula.
- Outputs elements include the ratio between the free receptor and the baseline. This output on the regular grid is useful in the exploration. Simulation uses only the value at the end of the treatment. The latter choice speeds up the computations in case of many individuals and/or replicates.
Exploration
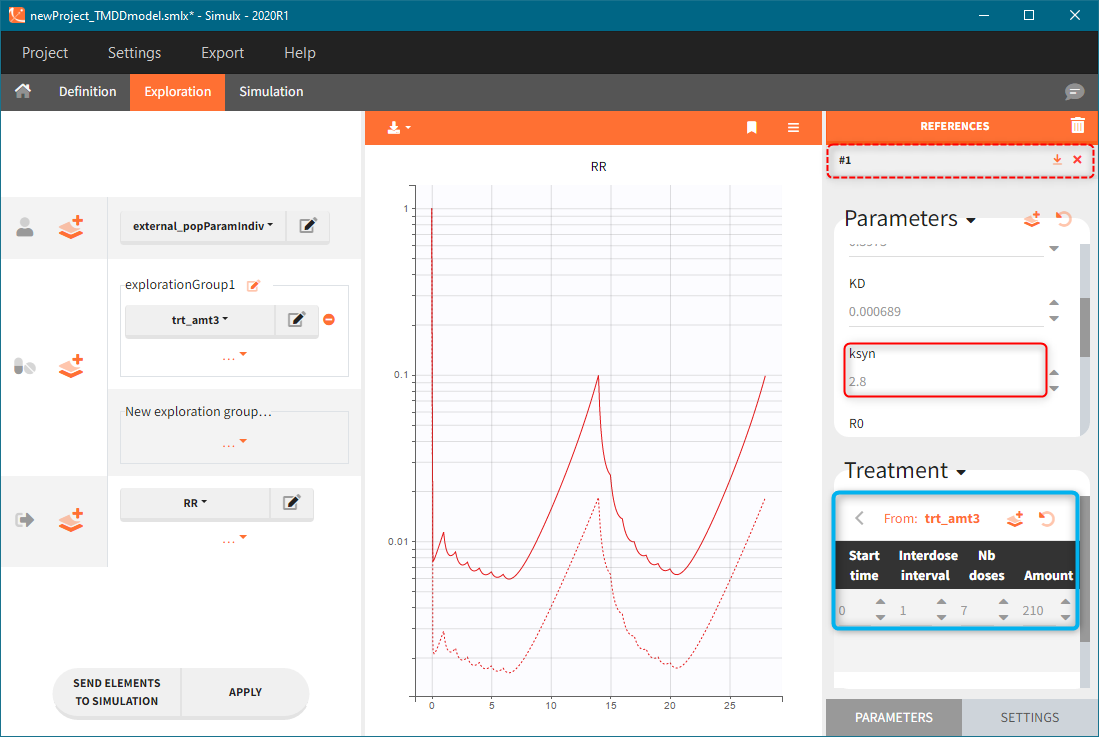
The model assumes that rheumatoid arthritis disease affects the synthesis of the free receptor (ksyn parameter). It means that for a certain dose level the efficacy target can be reached by healthy individuals (ksyn_pop = 0.789, dashed reference curve in the figure below), but not by patients with rheumatoid arthritis (ksyn_pop = 2.8, solid curve). In this exploration, the scaling of the amount 3mg/kg corresponds to typical individual with a weight 70kg (blue frame).
Simulation
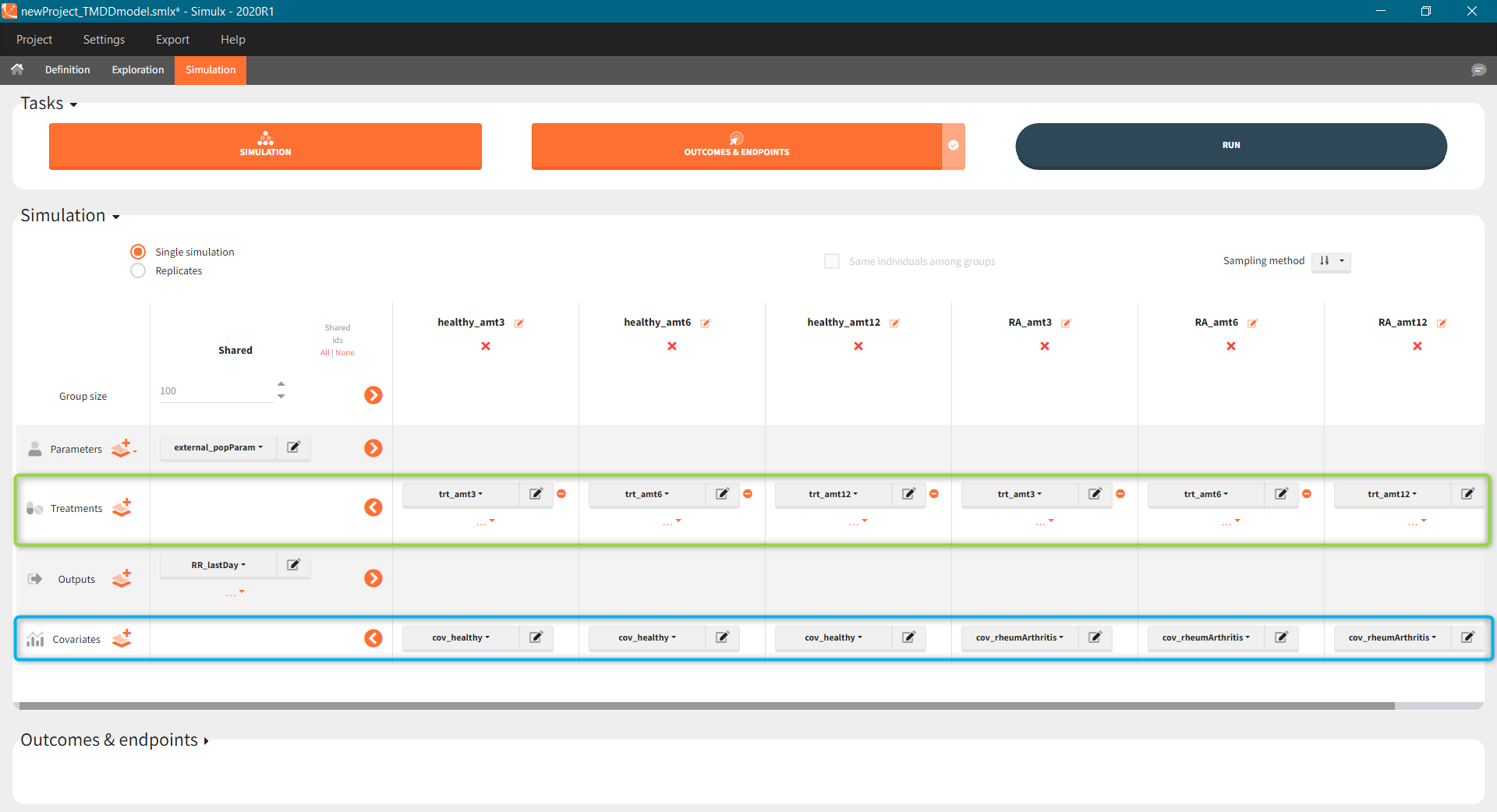
The scenario consists of 6 groups for three dose levels and two populations, see here to learn about creating a simulation scenario.
- Treatment is group specific. Each group uses one of the three covariate dependent doses: 3mg/kg, 6mg/kg or 12mg/kg (green frame).
- Type of a population – healthy or with the rheumatoid arthritis – is in the group specific covariate (blue frame).
- Group size, population parameters and the output are the same for all groups.
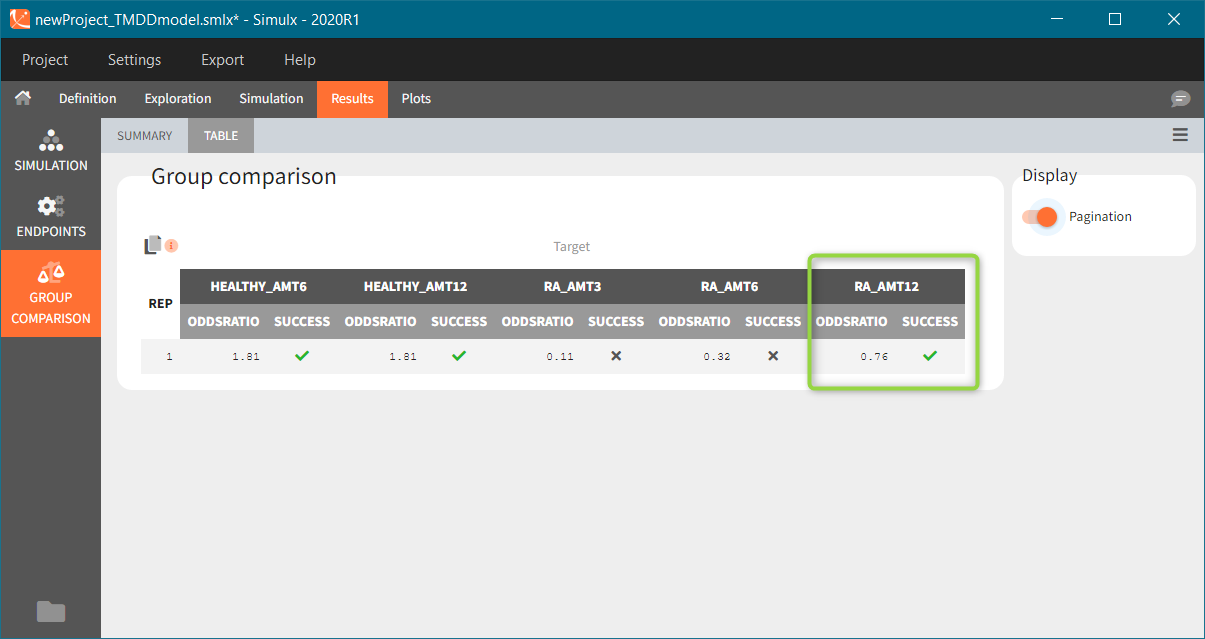
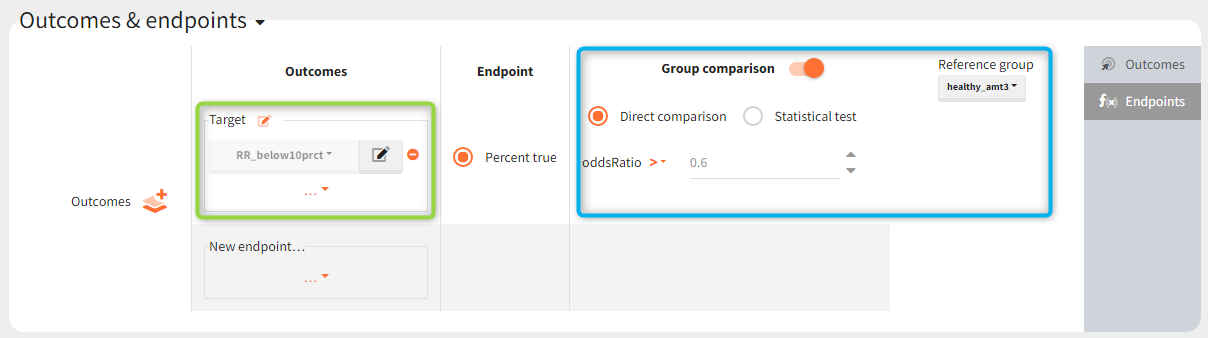
The outcomes&endpoints section defines post-processing of the simulation outputs for post-processing, which helps to compare the groups. The main endpoint is the percentage of individuals reaching the efficacy target in each group. It summarizes the “true” outcomes, which correspond to the situation when the ratio between the free receptor and the baseline at the end of the treatment is less then 10% (green frame). In addition, the “group comparison” (blue frame) compares the endpoints of all groups with respect to the reference group (healthy population with 3mg/kg dose level).
Results
To run the simulation and the calculation of the endpoints it is enough to select both tasks and click the button “Run”. Generation of the results and plots is automatic in two dedicated tabs. The outcome distribution shows that the efficacy of the treatment in healthy individuals stays above 90% (red line) for all dose levels. On the contrary, a group with rheumatoid arthritis achieves this level only at the highest tested dose.
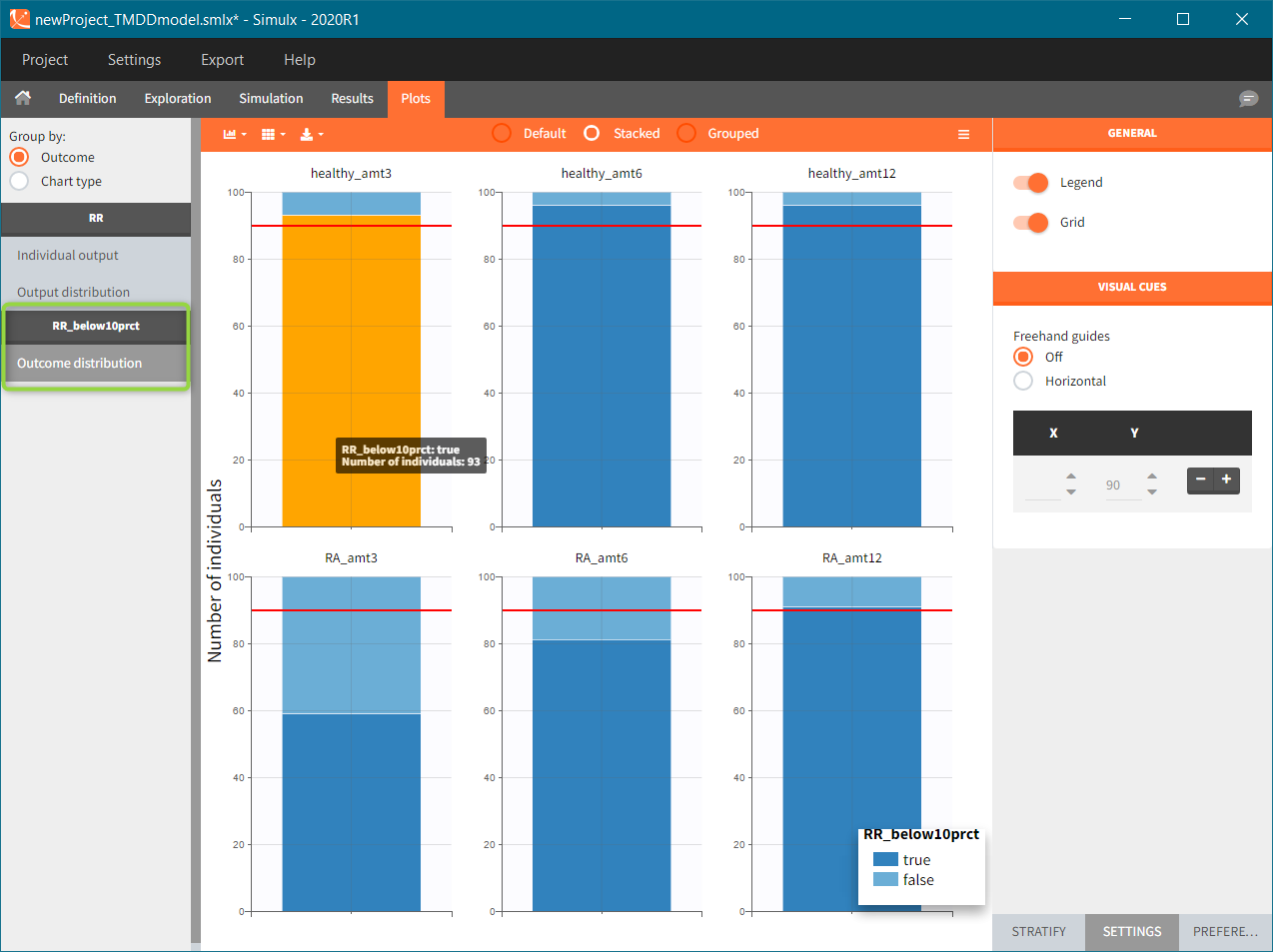
The response at the lowest dose level by healthy individuals is the reference. Group comparison considers a test group a “success” if the odds ratio of “percentage true” is larger then 0.6. Only the highest dose level satisfies this criterion for unhealthy patients.